Avian Influenza Virus (AIV)
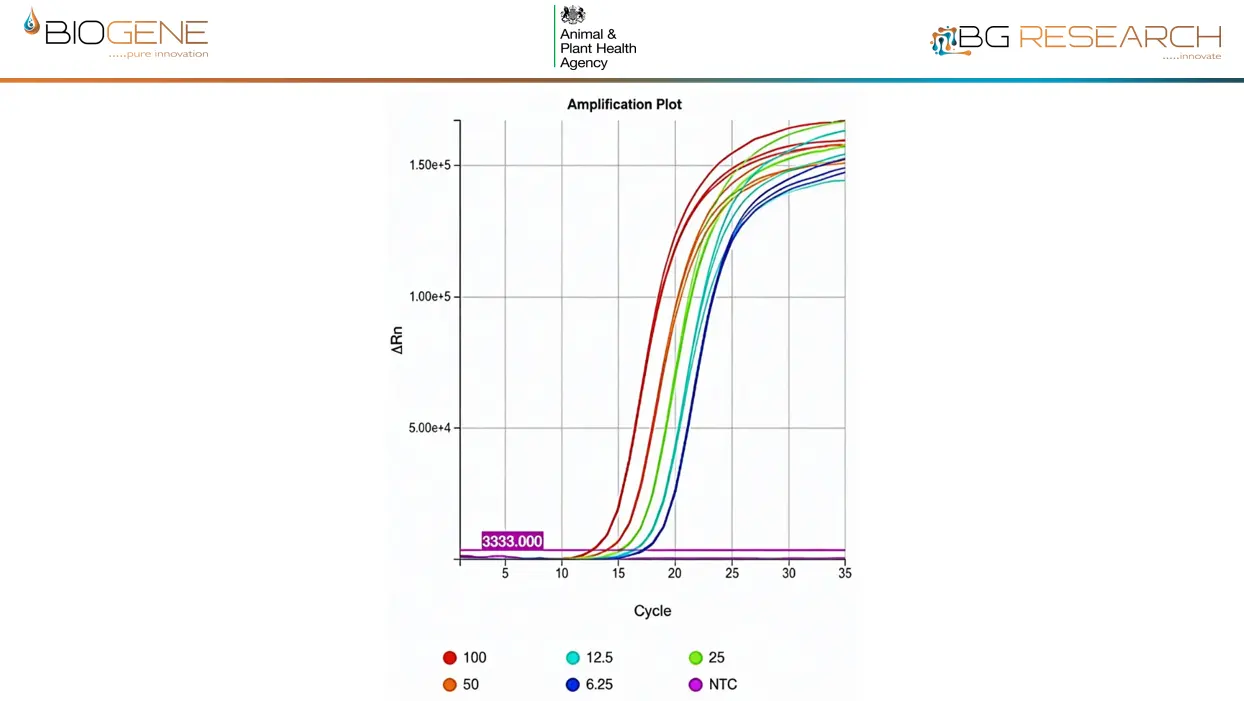
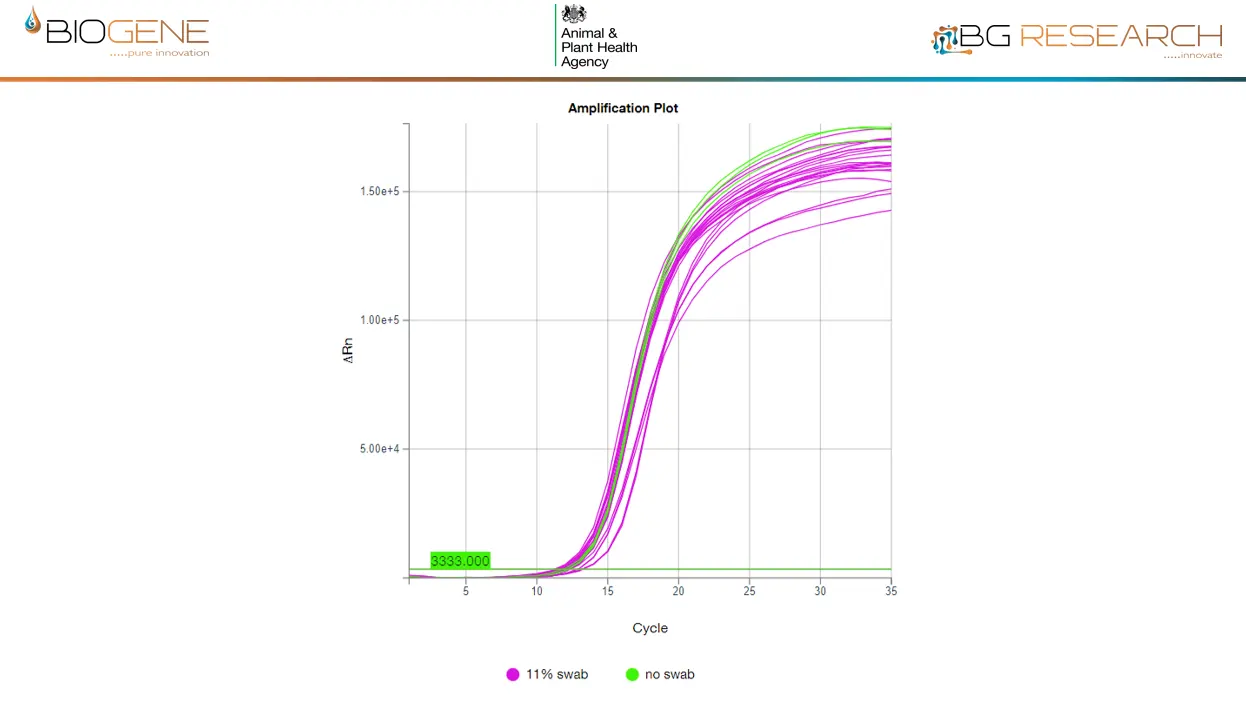
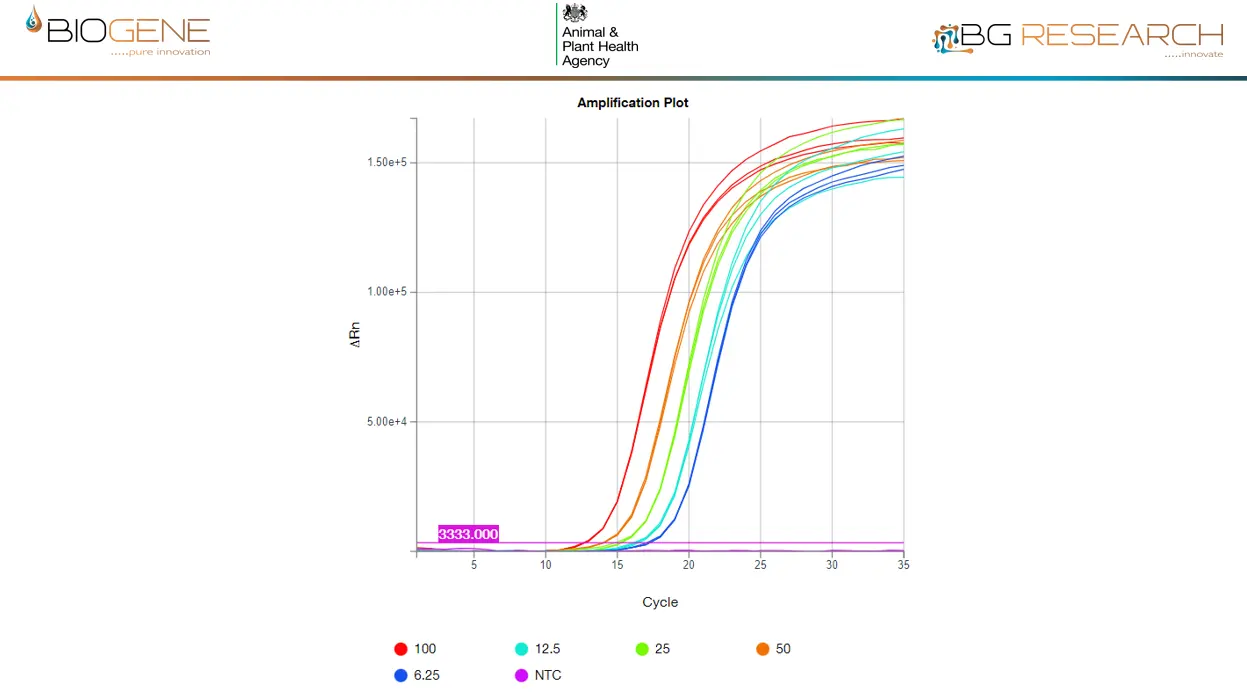
Direct detection of inactivated AIV of the current UK outbreak strain, spiked into claocal swab at 6-100 virions/rxn. LLOD is <1,000 virions/ml of eluate

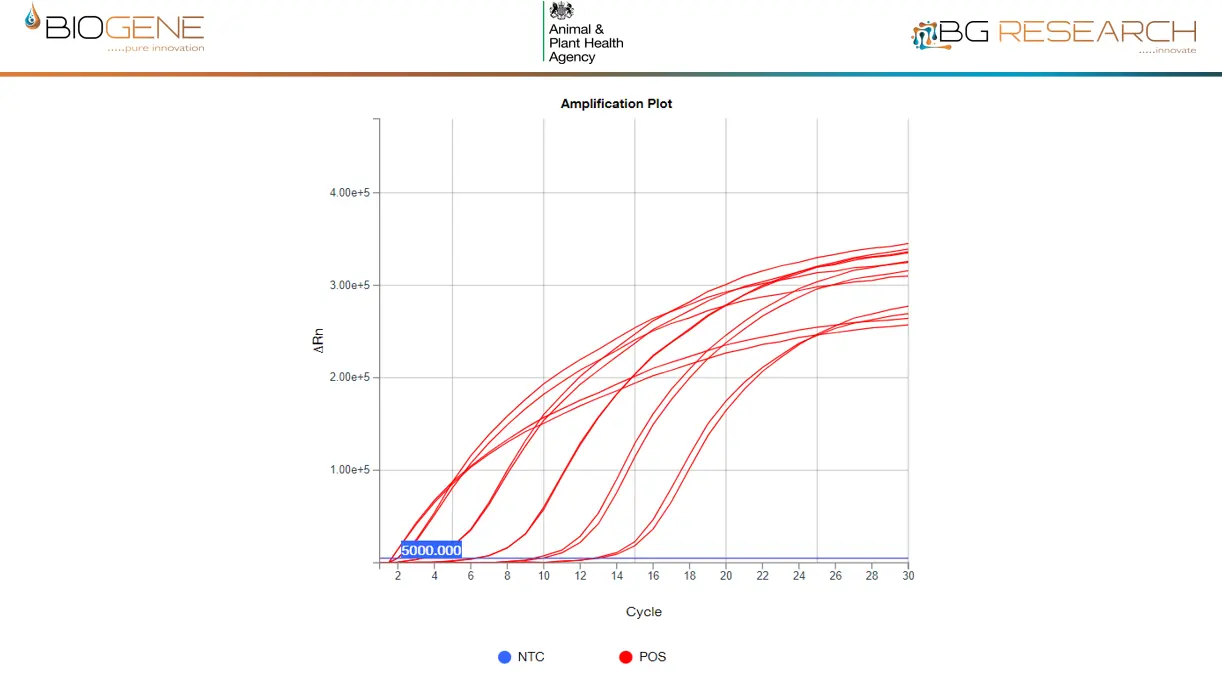
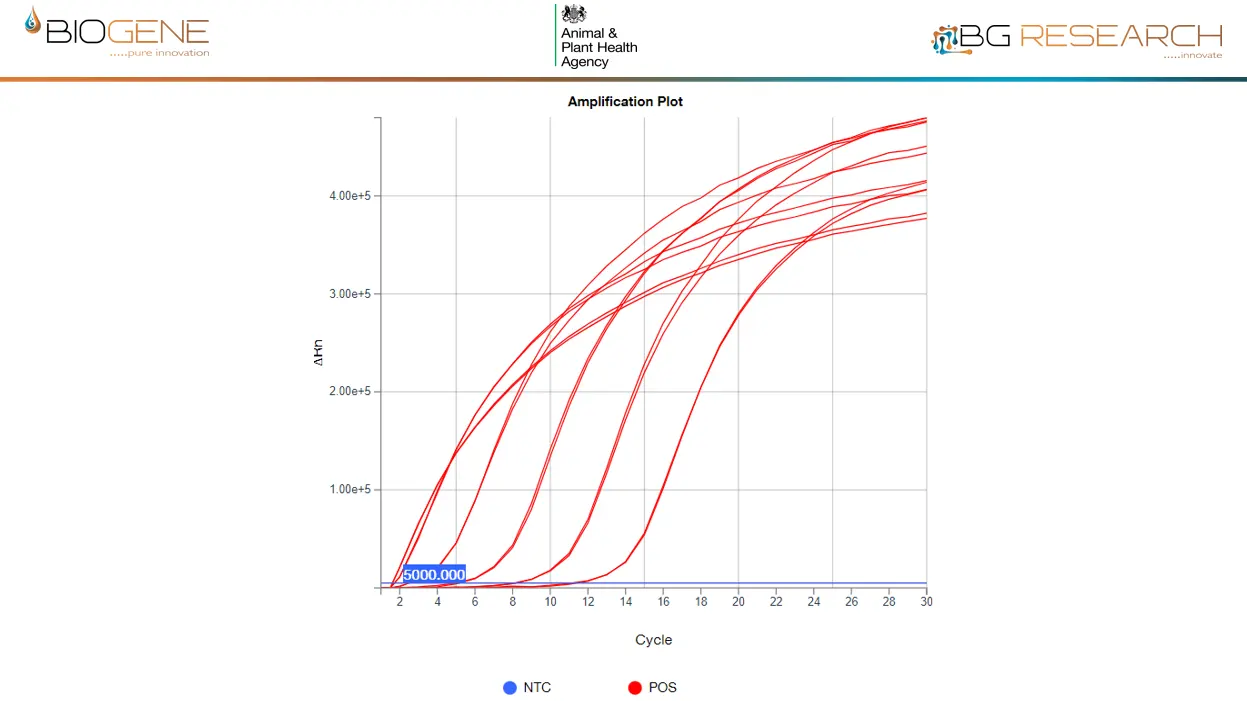
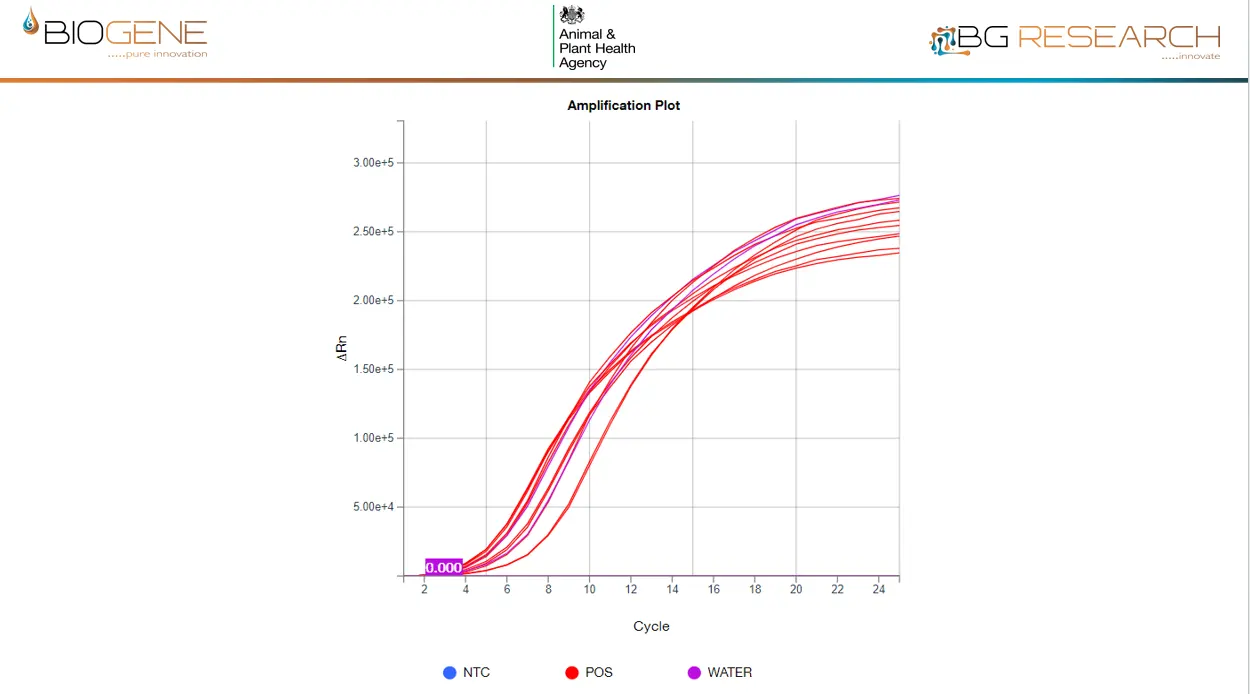
Detects >99.9% of Known AIV Strains Worldwide
The AIV assay is built on complete global sequence coverage over 7 years, using a comprehensive primer set and single universal probe.
- Sample type: Cloacal or oropharyngeal swabs
- Preparation: Elute into 1 ml water; add 16% v/v to reaction – no extraction
- LOD: <1,000 virions/ml
- Time to result: ~30 minutes
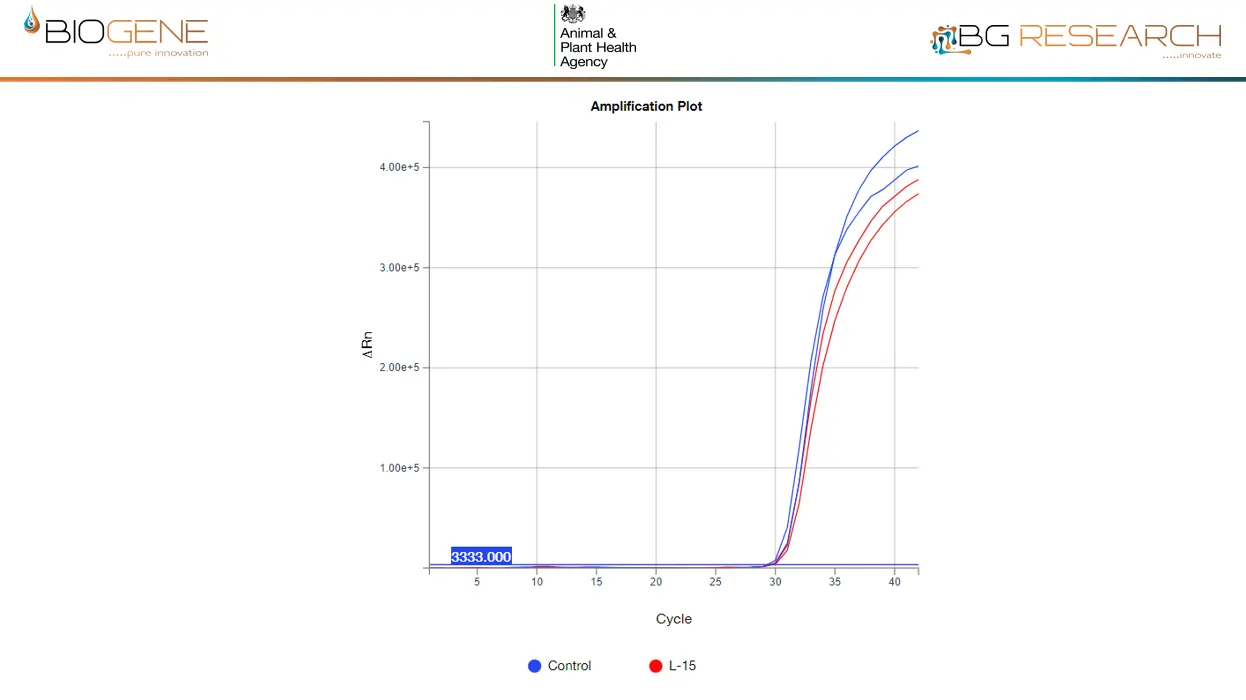
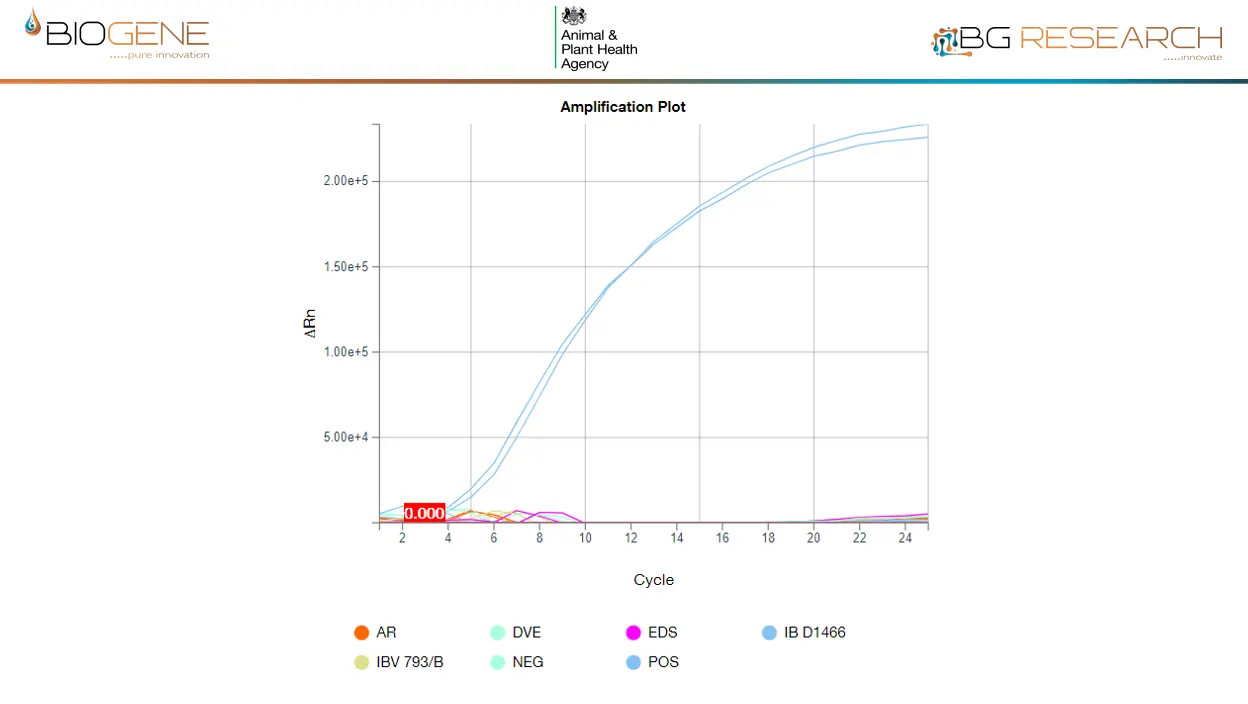
- Cross-reactivity: None observed across broad avian pathogen panel
Use for farm-side screening, import/export testing, or wildlife surveillance. H5 and H7 subtype-specific assays are in development.
Performance data generated through an Innovate UK funded research project, involving the Animal & Plant Health Agency (APHA), demonstrate the rapid molecular detection capability of the CENOS platform under defined study conditions. The participating institutions do not endorse the product.